CHEMICAL EQUILIBRIUM
Chemical equilibrium deals with extent to which a chemical reaction proceeds.
It is observed that most of the chemical reactions do not go to completion i.e., the reactants are not completely converted to products. The reaction proceeds to certain extent and reaches a state at which the concentrations of both reactants and products remain constant with time. This state is generally referred to as equilibrium state.
In these reactions, not only the conversion of reactants to products occurs, but also the conversion of products to reactants is also possible. These reactions are known as reversible reactions, which reach equilibrium state at which the number of reactant species converted to products becomes equal to the number of product species converted to reactants. That is why at equilibrium, there is no observable change in the concentration of reactants and products. The reaction is said to be halted and no further conversion of reactants is possible under given set of conditions.
Chemical equilibrium deals with these reversible reactions, which reach equilibrium state. The scope of chemical equilibrium includes the study of characteristics and factors affecting the chemical equilibria.
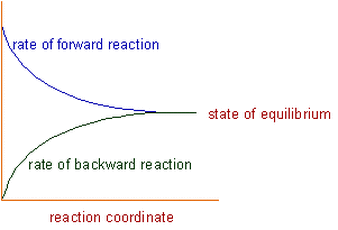
Chemical equilibrium: The state at which the rate of forward reaction becomes equal to the rate of backward reaction is called chemical equilibrium.
Explanation: Initially the rate of forward reaction is greater than the rate of backward reaction. However during the course of reaction, the concentration of reactants decreases and the concentration of products increases. Since the rate of a reaction is directly proportional to the concentration, the rate of forward reaction decreases with time, whereas the rate of backward reaction increases.
At certain stage, both the rates become equal. From this point onwards, there will be no change in the concentrations of both reactants and products with time. This state is called as equilibrium state.
The state of chemical equilibrium can be shown graphically in the picture above.
IRREVERSIBLE AND REVERSIBLE REACTIONS
Irreversible reaction: A reaction that occurs only in one direction is called an irreversible reaction i.e., only the reactants are converted to products and the conversion of products to reactants is not possible.
The single headed arrow is used to indicate the irreversible reactions.
E.g.
1) The combustion of methane is an irreversible reaction since it is not possible to convert the products (carbon dioxide and water) back to the reactants (methane and oxygen).
CH4 + 2O2 → CO2 + 2H2O
Reversible reaction : A reaction that occurs in both forward and backward directions is called reversible reaction. In a reversible reaction, the reactants are converted into products and the products can also be converted back to the reactants.
The half headed arrows are used to indicate the irreversible reactions.
H2 + I2 ↔ 2HI
The chemical equilibrium is possible in reversible reactions only.
TYPES OF CHEMICAL EQUILIBRIA
The chemical equilibria are classified into two types: 1) Homogeneous equilibrium and 2) Heterogeneous equilibrium.
1) Homogeneous equilibrium: A chemical equilibrium is said to be homogeneous if all the substances (reactants and products) at equilibrium are in the same phase.
2) Heterogeneous equilibrium: A chemical equilibrium is said to be heterogeneous if all the substances at equilibrium are not in the same phase.
-Christine Joy G. Jimenez
IV- Zara, Group 5
It is observed that most of the chemical reactions do not go to completion i.e., the reactants are not completely converted to products. The reaction proceeds to certain extent and reaches a state at which the concentrations of both reactants and products remain constant with time. This state is generally referred to as equilibrium state.
In these reactions, not only the conversion of reactants to products occurs, but also the conversion of products to reactants is also possible. These reactions are known as reversible reactions, which reach equilibrium state at which the number of reactant species converted to products becomes equal to the number of product species converted to reactants. That is why at equilibrium, there is no observable change in the concentration of reactants and products. The reaction is said to be halted and no further conversion of reactants is possible under given set of conditions.
Chemical equilibrium deals with these reversible reactions, which reach equilibrium state. The scope of chemical equilibrium includes the study of characteristics and factors affecting the chemical equilibria.
Chemical equilibrium: The state at which the rate of forward reaction becomes equal to the rate of backward reaction is called chemical equilibrium.
Explanation: Initially the rate of forward reaction is greater than the rate of backward reaction. However during the course of reaction, the concentration of reactants decreases and the concentration of products increases. Since the rate of a reaction is directly proportional to the concentration, the rate of forward reaction decreases with time, whereas the rate of backward reaction increases.
At certain stage, both the rates become equal. From this point onwards, there will be no change in the concentrations of both reactants and products with time. This state is called as equilibrium state.
The state of chemical equilibrium can be shown graphically in the picture above.
IRREVERSIBLE AND REVERSIBLE REACTIONS
Irreversible reaction: A reaction that occurs only in one direction is called an irreversible reaction i.e., only the reactants are converted to products and the conversion of products to reactants is not possible.
The single headed arrow is used to indicate the irreversible reactions.
E.g.
1) The combustion of methane is an irreversible reaction since it is not possible to convert the products (carbon dioxide and water) back to the reactants (methane and oxygen).
CH4 + 2O2 → CO2 + 2H2O
Reversible reaction : A reaction that occurs in both forward and backward directions is called reversible reaction. In a reversible reaction, the reactants are converted into products and the products can also be converted back to the reactants.
The half headed arrows are used to indicate the irreversible reactions.
H2 + I2 ↔ 2HI
The chemical equilibrium is possible in reversible reactions only.
TYPES OF CHEMICAL EQUILIBRIA
The chemical equilibria are classified into two types: 1) Homogeneous equilibrium and 2) Heterogeneous equilibrium.
1) Homogeneous equilibrium: A chemical equilibrium is said to be homogeneous if all the substances (reactants and products) at equilibrium are in the same phase.
2) Heterogeneous equilibrium: A chemical equilibrium is said to be heterogeneous if all the substances at equilibrium are not in the same phase.
-Christine Joy G. Jimenez
IV- Zara, Group 5
Monoprotic and Polyprotic Acids

What do you think is a Monoprotic acid and a Polyprotic acid? What do you think will their effect in a chemical reaction? So, let’s stop asking and do the learning. Here are some facts that we should know about these two acids.
Let’s discuss first about the Monoprotic acid. Monoprotic acids are those acids that are able to donate one proton per molecule during the process of dissociation (sometimes called ionization). An example of dissociation process is given below that is represented by (HA):
HA (aq) + H2O (l) ↔ H3O+ (aq) + A− (aq)
The example given explains that a monoprotic acid represented by the symbol (HA) donates one proton in the water that will produce a product of hydronium and anion. Common examples of monoprotic acids in mineral acids include hydrochloric acid (HCl) and nitric acid (HNO3). It can also be determine with only one acidic Hydrogen atom like the given examples of monoprotic acids. On the other hand, for organic acids the term mainly indicates the presence of one carboxylic acid group and sometimes these acids are known as monocarboxylic acid.
Now let’s further our knowledge about the Polyprotic acid. Polyprotic acids, also known as polybasic acids, are able to donate more than one proton per acid molecule, in contrast to monoprotic acids that only donate one proton per molecule. Specific types of polyprotic acids have more specific names, such as diprotic acid that have a potential to donate two protons and triprotic acid that can donate three protons.
The example below will show how a diprotic acid donates in the reaction:
H2A (aq) + H2O (l) ↔ H3O+ (aq) + HA− (aq)
HA− (aq) + H2O (l) ↔ H3O+ (aq) + A2− (aq)
The diprotic acid (H2A(aq)) undergone two reaction because of being capable of donating two protons with other molecules. Some examples of diprotic acids like sulfuric acid (H2SO4), carbonic acid (H2CO3), hydrogen sulfide (H2S), chromic acid (H2CrO4), and oxalic acid (H2C2O4) can also undergo to hat kind of process in the example. We can also notice that a diprotic acid have two acidic Hydrogen atoms.
Here is another example for a tripotic acid:
H3A (aq) + H2O (l) ↔ H3O+(aq) + H2A−(aq)
H2A− (aq) + H2O (l) ↔ H3O+ (aq) + HA2− (aq)
HA2− (aq) + H2O (l) ↔ H3O+ (aq) + A3− (aq)
H3A in aqueous solution is the symbolical representation for a tripotic acid that shows three different dissociation processes that donates three protons, the same thing with the monoprotic and diprotic but with three protons to donate with other molecules. Tripotic acids are also distinguished with the presence of three acidic hydrogen atoms like phosphoric acid (H3PO4) and citric acid (C6H8O7).
- Donna Clarisse L. Calma
IV-Zara, Group 5
Let’s discuss first about the Monoprotic acid. Monoprotic acids are those acids that are able to donate one proton per molecule during the process of dissociation (sometimes called ionization). An example of dissociation process is given below that is represented by (HA):
HA (aq) + H2O (l) ↔ H3O+ (aq) + A− (aq)
The example given explains that a monoprotic acid represented by the symbol (HA) donates one proton in the water that will produce a product of hydronium and anion. Common examples of monoprotic acids in mineral acids include hydrochloric acid (HCl) and nitric acid (HNO3). It can also be determine with only one acidic Hydrogen atom like the given examples of monoprotic acids. On the other hand, for organic acids the term mainly indicates the presence of one carboxylic acid group and sometimes these acids are known as monocarboxylic acid.
Now let’s further our knowledge about the Polyprotic acid. Polyprotic acids, also known as polybasic acids, are able to donate more than one proton per acid molecule, in contrast to monoprotic acids that only donate one proton per molecule. Specific types of polyprotic acids have more specific names, such as diprotic acid that have a potential to donate two protons and triprotic acid that can donate three protons.
The example below will show how a diprotic acid donates in the reaction:
H2A (aq) + H2O (l) ↔ H3O+ (aq) + HA− (aq)
HA− (aq) + H2O (l) ↔ H3O+ (aq) + A2− (aq)
The diprotic acid (H2A(aq)) undergone two reaction because of being capable of donating two protons with other molecules. Some examples of diprotic acids like sulfuric acid (H2SO4), carbonic acid (H2CO3), hydrogen sulfide (H2S), chromic acid (H2CrO4), and oxalic acid (H2C2O4) can also undergo to hat kind of process in the example. We can also notice that a diprotic acid have two acidic Hydrogen atoms.
Here is another example for a tripotic acid:
H3A (aq) + H2O (l) ↔ H3O+(aq) + H2A−(aq)
H2A− (aq) + H2O (l) ↔ H3O+ (aq) + HA2− (aq)
HA2− (aq) + H2O (l) ↔ H3O+ (aq) + A3− (aq)
H3A in aqueous solution is the symbolical representation for a tripotic acid that shows three different dissociation processes that donates three protons, the same thing with the monoprotic and diprotic but with three protons to donate with other molecules. Tripotic acids are also distinguished with the presence of three acidic hydrogen atoms like phosphoric acid (H3PO4) and citric acid (C6H8O7).
- Donna Clarisse L. Calma
IV-Zara, Group 5
Lewis structures
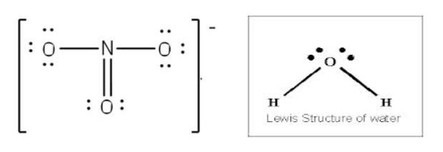
(also known as Lewis dot diagrams, electron dot diagrams, and electron dot structures) are diagrams that show the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule. A Lewis structure can be drawn for any covalently bonded molecule, as well as coordination compounds. The Lewis structure was named after Gilbert Newton Lewis, who introduced it in his 1916 article The Atom and the Molecule. They are similar to electron dot diagrams in that the valence electrons in lone pairs are represented as dots, but they also contain lines to represent shared pairs in a chemical bond (single, double, triple, etc.)
Steps to Drawing a Lewis Structure
1. Pick a Central Atom
Start your structure by picking a central atom and writing its element symbol. This atom will be the one with the lowest electronegativity. Sometimes it's difficult to know which atom is the least electronegative, but you can use the periodic table trends to help you out. Electronegativity typically increases as you move from left to right across the periodic table and decreases as you move down the table, from top to bottom.
2. Count Electrons
Lewis electron dot structures show the valence electrons for each atom. You don't need to worry about the total number of electrons, only those in the the outer shells. The octet rule states that atoms with 8 electrons in their outer shell are stable.
3. Place Electrons around Atoms
Once you have determined how many electrons to draw around each atom, start placing them on the structure. Start by placing one pair of dots for each pair of valence electrons. Once the lone pairs are placed, you may find some atoms, particularly the central atom, don't have a complete octet of electrons. This indicates there are double or possibly triple bonds.
Once the electrons have been placed, put brackets around the entire structure. If there is a charge on the molecule, write it as a superscript on the upper right, outside of the bracket.
-Matthew W. Garcia
IV-Zara, Group 5
Steps to Drawing a Lewis Structure
1. Pick a Central Atom
Start your structure by picking a central atom and writing its element symbol. This atom will be the one with the lowest electronegativity. Sometimes it's difficult to know which atom is the least electronegative, but you can use the periodic table trends to help you out. Electronegativity typically increases as you move from left to right across the periodic table and decreases as you move down the table, from top to bottom.
2. Count Electrons
Lewis electron dot structures show the valence electrons for each atom. You don't need to worry about the total number of electrons, only those in the the outer shells. The octet rule states that atoms with 8 electrons in their outer shell are stable.
3. Place Electrons around Atoms
Once you have determined how many electrons to draw around each atom, start placing them on the structure. Start by placing one pair of dots for each pair of valence electrons. Once the lone pairs are placed, you may find some atoms, particularly the central atom, don't have a complete octet of electrons. This indicates there are double or possibly triple bonds.
Once the electrons have been placed, put brackets around the entire structure. If there is a charge on the molecule, write it as a superscript on the upper right, outside of the bracket.
-Matthew W. Garcia
IV-Zara, Group 5
Hybridization of Atomic Orbitals
If the four hydrogen atoms in a methane molecule (CH4) were bound to the three 2p orbitals and the 2s orbital of the carbon atom, the H-C-H bond angles would be 90o for 3 of the hydrogen atoms and the 4th hydrogen atom would be at 135o from the others. Experimental evidence has shown that the bond angles in methane are not arranged that way but are 109.5o giving the overall shape of a tetrahedron. The tetrahedral structure makes much more sense in that hydrogen atoms would naturally repel each other due to their negative electron clouds and form this shape. If you think electron-electron repulsion isn't significant, try walking through a wall! There is plenty of space for your nuclei to pass through the nuclei of the wall material but ouch, it just doesn't work that way.
Experimental evidence has also shown that the H-N-H bond angles in ammonia (NH3) are 107o and the H-O-H bond angles in water are 105o. It is clear from these bond angles that the non-bonding pairs of electrons occupy a reasonable amount of space and are pushing the hydrogen atoms closer together compared to the angles found in methane.
The valence shell electron-pair repulsion model (VESPR) was devised to account for these molecular shapes. In this model, atoms and pairs of electrons will be arranged to minimize the repulsion of these atoms and pairs of electrons. Since the non-bonded electron pairs are held somewhat closer to the nucleus than the attached hydrogen atoms, they tend to crowd the hydrogen atoms. Thus ammonia exists as a distorted tetrahedron (trigonal pyramidal) rather than a trigonal plane and water also exists as a distorted tetrahedron (bent) rather than a linear molecule with the hydrogen atoms at a 180o bond angle.
This concept proposes that since the attached groups are not at the angles of the p orbitals and their atomic orbitals would not have maximum overlap (to form strong bonds) the s and p orbitals will be hybridized to match the bond angles of the attached groups.
The number of these new hybrid orbitals must be equal to the numbers of atoms and non-bonded electron pairs surrounding the central atom! This valence shell repulsion model can be illustrated at home with a very fun experiment!
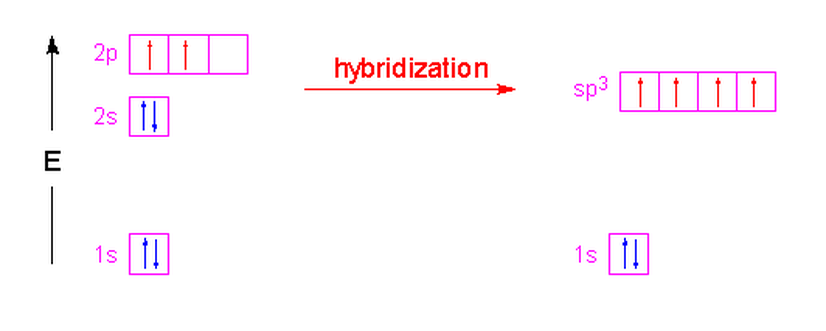
In the case of methane, the three 2p orbitals of the carbon atom are combined with its 2s orbital to form four new orbitals called "sp3" hybrid orbitals. The name is simply a tally of all the orbitals that were blended together to form these new hybrid orbitals. Four hybrid orbitals were required since there are four atoms attached to the central carbon atom. These new orbitals will have an energy slightly above the 2s orbital and below the 2p orbitals as shown in the following illustration. Notice that no change occurred with the 1s orbital.
These hybrid orbitals have 75% p-character and 25% s-character which gives them a shape that is shorter and fatter than a p-orbital. The new shape looks a little like...
A stick and wedge drawing of methane shows the tetrahedral angles...(The wedge is coming out of the paper and the dashed line is going behind the paper. The solid lines are in the plane of the paper.)
A space-filling model of methane would look like...
In the case of ammonia, the three 2p orbitals of the nitrogen atom are combined with the 2s orbital to form four sp3 hybrid orbitals. The non-bonded electron pair will occupy a hybrid orbital. Again we need a hybrid orbital for each atom and pair of non-bonding electrons. Ammonia has three hydrogen atoms and one non-bonded pair of electrons when we draw the electron-dot formula. In order to determine the hybridization of an atom, you must first draw the electron-dot formula.
A stick and wedge drawing of ammonia showing the non-bonding electrons in a probability area for the hybrid orbital...
A space-filling model of ammonia would look like...(Note the non-bonded electron pair is not shown in this model.)
In the case of water, the three 2p orbitals of the oxygen atom are combined with the 2s orbital to form four sp3 hybrid orbitals. The two non-bonded electron pairs will occupy hybrid orbitals. Again we need a hybrid orbital for each atom and each pair of non-bonding electrons. Water has two hydrogen atoms and two non-bonded pairs of electrons when we draw the electron-dot formula.
A stick and wedge drawing of water showing the non-bonding electron pairs in probability areas for the hybrid orbital...
A space-filling model of water would look like...(Note the non-bonded electron pairs are not shown in this model.)
Now let's look at something a bit different. In the boron trifluoride molecule, only three groups are arranged around the central boron atom. In this case, the 2s orbital is combined with only two of the 2p orbitals (since we only need three hybrid orbitals for the three groups...thinking of groups as atoms and non-bonding pairs) forming three hybrid orbitals called sp2 hybrid orbitals. The other p-orbital remains unhybridized and is at right angles to the trigonal planar arrangement of the hybrid orbitals. The trigonal planar arrangement has bond angles of 120o.
In the following stick model, the empty p orbital is shown as the probability area...one end shaded blue and the other is white...there are no electrons in this orbital!
A space-filling model of boron trifluoride would look like...
Finally let's look at beryllium dichloride. Since only two groups are attached to beryllium, we only will have two hybrid orbitals. In this case, the 2s orbital is combined with only one of the 2p orbitals to yield two sp hybrid orbitals. The two hybrid orbitals will be arranged as far apart as possible from each other with the result being a
linear arrangement. The two unhybridized p-orbitals stay in their respective positions (at right angles to each other) and perpendicular to the linear molecule.
In the following stick model, the empty p orbitals are shown as the probability areas...one green and one blue.
A space-filling model of beryllium dichloride would look like...
Hybridization Involving d-Orbitals
As we discussed earlier, some 3rd row and larger elements can accommodate more than eight electrons around the central atom. These atoms will also be hybridized and have very specific arrangements of the attached groups in space. The two types of hybridization involved with d orbitals are sp3d and sp3d2.
The groups will be arranged in a trigonal bipyramidal arrangement with sp3d hybridization...bond angles will be 120o in the plane with two groups arranged vertically above and below this plane.
There will be an octahedral arrangement with sp3d2 hybridization...all bond angles are at 90o.
Non-bonded electron pairs are always placed where they will have the most space...in the trigonal plane for sp3d hybridization.
Try drawing the 3-dimensional electron-dot picture for each of the following molecules...First draw an electron-dot formula. Remember to put all the extra electrons on the central atom as pairs when drawing this initial electron-dot formula. Now count the groups around the central atom. If there are six groups (Remember to count non-bonding electron pairs as groups.) it will have sp3d2 hybridization. If it has five groups it will have sp3d hybridization. SF6 sulfur hexafluoride PF5 phosphorus pentafluoride SF4 sulfur tetrafluoride ClF3 chlorine trifluoride XeF2 xenon difluoride
- Crissa Chirene P. Bug-os
IV-Zara, Group 5
Experimental evidence has also shown that the H-N-H bond angles in ammonia (NH3) are 107o and the H-O-H bond angles in water are 105o. It is clear from these bond angles that the non-bonding pairs of electrons occupy a reasonable amount of space and are pushing the hydrogen atoms closer together compared to the angles found in methane.
The valence shell electron-pair repulsion model (VESPR) was devised to account for these molecular shapes. In this model, atoms and pairs of electrons will be arranged to minimize the repulsion of these atoms and pairs of electrons. Since the non-bonded electron pairs are held somewhat closer to the nucleus than the attached hydrogen atoms, they tend to crowd the hydrogen atoms. Thus ammonia exists as a distorted tetrahedron (trigonal pyramidal) rather than a trigonal plane and water also exists as a distorted tetrahedron (bent) rather than a linear molecule with the hydrogen atoms at a 180o bond angle.
This concept proposes that since the attached groups are not at the angles of the p orbitals and their atomic orbitals would not have maximum overlap (to form strong bonds) the s and p orbitals will be hybridized to match the bond angles of the attached groups.
The number of these new hybrid orbitals must be equal to the numbers of atoms and non-bonded electron pairs surrounding the central atom! This valence shell repulsion model can be illustrated at home with a very fun experiment!
In the case of methane, the three 2p orbitals of the carbon atom are combined with its 2s orbital to form four new orbitals called "sp3" hybrid orbitals. The name is simply a tally of all the orbitals that were blended together to form these new hybrid orbitals. Four hybrid orbitals were required since there are four atoms attached to the central carbon atom. These new orbitals will have an energy slightly above the 2s orbital and below the 2p orbitals as shown in the following illustration. Notice that no change occurred with the 1s orbital.
These hybrid orbitals have 75% p-character and 25% s-character which gives them a shape that is shorter and fatter than a p-orbital. The new shape looks a little like...
A stick and wedge drawing of methane shows the tetrahedral angles...(The wedge is coming out of the paper and the dashed line is going behind the paper. The solid lines are in the plane of the paper.)
A space-filling model of methane would look like...
In the case of ammonia, the three 2p orbitals of the nitrogen atom are combined with the 2s orbital to form four sp3 hybrid orbitals. The non-bonded electron pair will occupy a hybrid orbital. Again we need a hybrid orbital for each atom and pair of non-bonding electrons. Ammonia has three hydrogen atoms and one non-bonded pair of electrons when we draw the electron-dot formula. In order to determine the hybridization of an atom, you must first draw the electron-dot formula.
A stick and wedge drawing of ammonia showing the non-bonding electrons in a probability area for the hybrid orbital...
A space-filling model of ammonia would look like...(Note the non-bonded electron pair is not shown in this model.)
In the case of water, the three 2p orbitals of the oxygen atom are combined with the 2s orbital to form four sp3 hybrid orbitals. The two non-bonded electron pairs will occupy hybrid orbitals. Again we need a hybrid orbital for each atom and each pair of non-bonding electrons. Water has two hydrogen atoms and two non-bonded pairs of electrons when we draw the electron-dot formula.
A stick and wedge drawing of water showing the non-bonding electron pairs in probability areas for the hybrid orbital...
A space-filling model of water would look like...(Note the non-bonded electron pairs are not shown in this model.)
Now let's look at something a bit different. In the boron trifluoride molecule, only three groups are arranged around the central boron atom. In this case, the 2s orbital is combined with only two of the 2p orbitals (since we only need three hybrid orbitals for the three groups...thinking of groups as atoms and non-bonding pairs) forming three hybrid orbitals called sp2 hybrid orbitals. The other p-orbital remains unhybridized and is at right angles to the trigonal planar arrangement of the hybrid orbitals. The trigonal planar arrangement has bond angles of 120o.
In the following stick model, the empty p orbital is shown as the probability area...one end shaded blue and the other is white...there are no electrons in this orbital!
A space-filling model of boron trifluoride would look like...
Finally let's look at beryllium dichloride. Since only two groups are attached to beryllium, we only will have two hybrid orbitals. In this case, the 2s orbital is combined with only one of the 2p orbitals to yield two sp hybrid orbitals. The two hybrid orbitals will be arranged as far apart as possible from each other with the result being a
linear arrangement. The two unhybridized p-orbitals stay in their respective positions (at right angles to each other) and perpendicular to the linear molecule.
In the following stick model, the empty p orbitals are shown as the probability areas...one green and one blue.
A space-filling model of beryllium dichloride would look like...
Hybridization Involving d-Orbitals
As we discussed earlier, some 3rd row and larger elements can accommodate more than eight electrons around the central atom. These atoms will also be hybridized and have very specific arrangements of the attached groups in space. The two types of hybridization involved with d orbitals are sp3d and sp3d2.
The groups will be arranged in a trigonal bipyramidal arrangement with sp3d hybridization...bond angles will be 120o in the plane with two groups arranged vertically above and below this plane.
There will be an octahedral arrangement with sp3d2 hybridization...all bond angles are at 90o.
Non-bonded electron pairs are always placed where they will have the most space...in the trigonal plane for sp3d hybridization.
Try drawing the 3-dimensional electron-dot picture for each of the following molecules...First draw an electron-dot formula. Remember to put all the extra electrons on the central atom as pairs when drawing this initial electron-dot formula. Now count the groups around the central atom. If there are six groups (Remember to count non-bonding electron pairs as groups.) it will have sp3d2 hybridization. If it has five groups it will have sp3d hybridization. SF6 sulfur hexafluoride PF5 phosphorus pentafluoride SF4 sulfur tetrafluoride ClF3 chlorine trifluoride XeF2 xenon difluoride
- Crissa Chirene P. Bug-os
IV-Zara, Group 5
