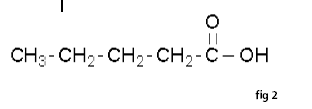CARBON AND LIFE
The term "organic" often invokes up the image of life; something very isolated from lifeless or not possessing any traits generally recognized to being alive. That’s why when we see that something is alive, therefore it is organic. In the supermarket, we are used to seeing the "organic" label to entitle something unusual, something more linked to nature. However, the term organic in chemistry has a very specific definition involving chemicals and chemistry that involves carbon. Organic chemistry is the study of carbon and the study of the chemistry of life. Since not all carbon reactions are organic, another way to look at organic chemistry would be to consider it the study of molecules containing the carbon-hydrogen bond and their reactions. It is a highly creative science. Chemists in general and organic chemists in particular can create new molecules never before proposed which, if carefully designed, may have important properties for the progress of the human understanding. Organic chemistry is important because it is the study of life and all of the chemical responses interconnected to life. Organic chemistry plays a part in the development of common household chemicals, medicine, merchandises, cosmetics and even human life. It can be truly concluded that most of the chemicals are part of daily life. Organic chemistry is interrelated to organic compounds.
Organic compounds may be classified in multiple ways. One major distinction is between natural and synthetic compounds. Natural compounds refer to those that are produced by plants or animals. Many of these are still extracted from natural sources because they would be far too expensive to be produced artificially. Examples include most sugars, certain nutrients such as vitamin B12, and, in general, those natural products with large or complicated molecules present in reasonable concentrations in living organisms. The other type is synthetic. Compounds that are prepared by reaction of other compounds are referred to as "synthetic". They may be either compounds that are not already established in plants or animals or those that do not occur naturally.
Organic substances are pretty vital in our lives. All the major types of biological molecules are organic molecules, and they are required for life. Remember, the chair that you sit at is mostly made up of a polymer of many glucose molecules. Your foods are probably made with vinegar, which comprises of mostly acetic acid, and other ingredients. Organic chemistry allows us to learn about these molecules. Carbon is the single element that makes up most of life -- organic chemistry studies all carbon based compounds, therefore Organic chemistry studies life itself.
references:
wikipedia.org
http://chemwiki.ucdavis.edu
http://chemistry.about.com
http://portal.acs.org
-Leomar T. Santiago
IV-Zara, Group5
Organic compounds may be classified in multiple ways. One major distinction is between natural and synthetic compounds. Natural compounds refer to those that are produced by plants or animals. Many of these are still extracted from natural sources because they would be far too expensive to be produced artificially. Examples include most sugars, certain nutrients such as vitamin B12, and, in general, those natural products with large or complicated molecules present in reasonable concentrations in living organisms. The other type is synthetic. Compounds that are prepared by reaction of other compounds are referred to as "synthetic". They may be either compounds that are not already established in plants or animals or those that do not occur naturally.
Organic substances are pretty vital in our lives. All the major types of biological molecules are organic molecules, and they are required for life. Remember, the chair that you sit at is mostly made up of a polymer of many glucose molecules. Your foods are probably made with vinegar, which comprises of mostly acetic acid, and other ingredients. Organic chemistry allows us to learn about these molecules. Carbon is the single element that makes up most of life -- organic chemistry studies all carbon based compounds, therefore Organic chemistry studies life itself.
references:
wikipedia.org
http://chemwiki.ucdavis.edu
http://chemistry.about.com
http://portal.acs.org
-Leomar T. Santiago
IV-Zara, Group5
Functional Groups
Functional groups are groups of atoms found within molecules that are involved in the chemical reactions characteristic of those molecules. These can pertain to any molecules, but but you are going to know more about them in organic chemistry. in their formulas, there are legends to be considered. R, R’,R’’… are considered as the attached hydrogen or hydrocarbon side chain or sometimes any group of atoms. C as Carbon and O as Oxygen.
It is also a portion of a molecule that is a recognizable/classified group of bound atoms. In organic chemistry it is very common to see molecules comprised mainly of a carbon backbone with functional groups attached to the chain. The functional group gives the molecule its properties, regardless of what molecule contains it*; they are centers of chemical reactivity. The functional groups within a molecule need to be identified when naming.
These are some of the examples of the Functional groups that you can usually encounter in Organic Chemistry
Functional Groups
1. Alkane-Hydrocarbons which contain only single bonds are called alkanes. They are called saturated hydrocarbons because there is hydrogen in every possible location. This gives them a general formula CnH2n+2.
2. Alkene-An alkene is a molecule made up entirely of carbon and hydrogen where on or more carbon atoms are connected by double bonds. The general formula for an alkene is CnH2n where n is the number of carbon atoms in the molecule.
3. Alkyne-The alkynes are the third homologous series of organic compounds of hydrogen and carbon, where there is at least one triple-bond between the atoms in the molecules.
4. Alkyl Halides -Alkyl halides [halo alkanes] consist of an alkyl group attached to a halogen:F, Cl, Br, I. Chloro, bromo and iodo alkyl halides are often susceptible to elimination and/or nucleophilic substitution reactions.
5. Aldehydes-Aldehydes have a hydrogen and an alkyl (or aromatic) group attached to a carbonyl function.Aldehydes can be shown in text as: RCHO
6. Ketones-Ketones have a pair of alkyl or aromatic groups attached to a carbonyl function. Ketones can be shown in text as: RCOR
7. Primary Alcohol-Primary alcohols have an -OH function attached to an R-CH2- group.Primary alcohols can be oxidised to aldehydes and on to carboxylic acids. (It can be difficult to stop the oxidation at the aldehyde stage.)Primary alcohols can be shown in text as: RCH2OH
8. Amine-In amines, the hydrogen atoms in the ammonia have been replaced one at a time by hydrocarbon groups. On this page, we are only looking at cases where the hydrocarbon groups are simple alkyl groups.
9. Amide-Primary amides (shown) have an alkyl or aromatic group attached to an amino-carbonyl function. Primary amides can be shown in text as: RCONH2. Secondary amides have an alkyl or aryl group attached to the nitrogen: RCONHR. Tertiary amides have two alkyl or aryl group attached to the nitrogen: RCONR2
10. Ether-Ethers have a pair of alkyl or aromatic groups attached to a linking oxygen atom.Ethers can be shown in text as: ROR
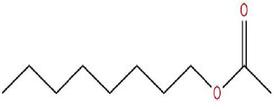
11. Ester-Esters are derived from carboxylic acids. A carboxylic acid contains the -COOH group, and in an ester the hydrogen in this group is replaced by a hydrocarbon group of some kind.
12. Nitrile-Nitriles (or organo cyanides) have an alkyl (or aromatic) group attached to a carbon-triple-bond-nitrogen function.Nitriles can be shown in text as: RCN
13. Acid Chlorides-Acid chlorides, or acyl chlorides, have an alkyl (or aromatic) group attached to a carbonyl function plus a labile (easily displaced) chlorine.Acid chlorides highly reactive entities are highly susceptible to attack by nucleophiles.Acid chlorides can be shown in text as: R-COCl or RCOCl.
14. Carboxylic Acid-Carboxylic acids have an alkyl or aromatic groups attached to a hydroxy-carbonyl function. Carboxylic acids can be shown in text as: RCOOH.
These functional groups are usually found in the substances we usually use in our daily lives particularly in our home. Alcohol, make-ups, Moth balls, Nail Polish, cooking oil, shampoo and even in the chips that you usually eat when you are going to have a movie marathon. In the invasion of the technology, these are used usually as an alternative. Functional groups are very useful in lives of everyone, but they are not aware of it.
REFERENCES:
http://www.sdsc.edu
http://chemmaster.co.in
http://www.worldofmolecules.com/
http://www.chemicalformula.org/
http://www.elmhurst.edu
http://www.ucc.ie
http://chemistry.about.com
http://www.chem.ucla.edu
-Jean Ellice P. Roy
Group 5 IV Zara
Nomenclature of Aromatic Hydrocarbons
|
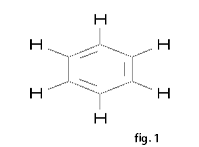
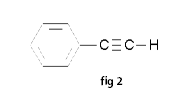
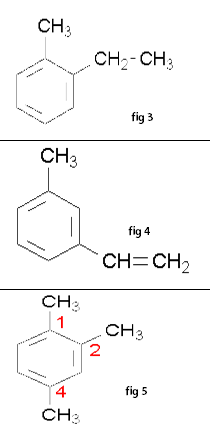
Benzene is the most common aromatic parent structure. a. The benzene ring (see fig 1) is named as a phenyl group when it is a substituent. Phenylethyne(see fig 2) b. Multiple substituents on a benzene ring are numbered to give these substituents the lowest possible numbers. When only two substituents are attached to a benzene ring, they can be named by the common nomenclature using ortho (o-) (1-2 placement), meta (m-) (1-3 placement) or para (p-) (1-4 placement). Examples: 1-ethyl-2-methylbenzene (see fig 3) o-methylethylbenzene (see fig 3) 1-ethenyl-3-methylbenzene (see fig 4) m-methylethenylbenzene (see fig 4) 1,2,4-trimethylbenzene (see fig 5) -Crissa Chirene P. Bug-os IV-Zara, Group5 |
ALKANE

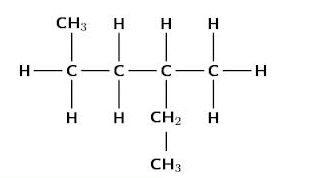
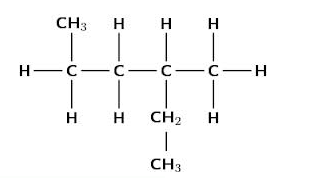
A saturated hydrocarbon or alkane is a hydrocarbon in which all of the carbon-carbon bonds are single bonds. The general formula for an alkane is CnH2n+2 where n is the number of carbon atoms in the molecule. There are two ways of writing a condensed structural formula. For example, butane may be written as CH3 CH2 CH2 CH3 or CH3 (CH2)2 CH3.
Rules for naming alkanes
1. Parent name of the molecule is determined by the number of carbons in the longest chain.
2. In the case where two chains have the same number of carbons, the parent is the chain with the most substituents.
3. The carbons in the chain are numbered starting from the end nearest the first substituent.
4. In the case where there are substituents having the same number of carbons from both ends, numbering starts from the end nearest the next substituent.
5. When more than one of a given substituent is present, a prefix is applied to indicate the number of substituents. Use di- for two, tri- for three, tetra- for four, etc. and use the number assigned to the carbon to indicate the position of each substituent.
Branched Alkanes
1. Count the number of carbons in the longest chain of the substituent.
2. Numbering of the substituent chain starts from the carbon attached to the parent chain.
3. Substituents are listed in alphabetical order. To alphabetize, ignore numerical (di-, tri-, tetra-) prefixes (e.g., ethyl would come before dimethyl).
Cyclic Alkanes
1. The parent name is determined by the number of carbons in the largest ring (e.g., cycloalkane such as cyclopentane).
2. In the case where the ring is attached to a chain containing additional carbons, the ring is considered to be a substituent on the chain. A substituted ring that is a substituent on something else is named using the rules for branched alkanes.
3. The carbons of the ring are numbered such that the substituents are given the lowest possible numbers.
Straight Chain Alkanes
# Carbon Name Molecula Structural
Formula Formula
1 Methane CH4 CH4
2 Ethane C2H6 CH3CH3
3 Propane C3H8 CH3CH2CH3
4 Butane C4H10 CH3CH2CH2CH3
5 Pentane C5H12 CH3CH2CH2CH2CH3
6 Hexane C6H14 CH3(CH2)4CH3
7 Heptane C7H16 CH3(CH2)5CH3
8 Octane C8H18 CH3(CH2)6CH3
9 Nonane C9H20 CH3(CH2)7CH3
10 Decane C10H22 CH3(CH2)8CH3
Rules for naming alkanes
1. Parent name of the molecule is determined by the number of carbons in the longest chain.
2. In the case where two chains have the same number of carbons, the parent is the chain with the most substituents.
3. The carbons in the chain are numbered starting from the end nearest the first substituent.
4. In the case where there are substituents having the same number of carbons from both ends, numbering starts from the end nearest the next substituent.
5. When more than one of a given substituent is present, a prefix is applied to indicate the number of substituents. Use di- for two, tri- for three, tetra- for four, etc. and use the number assigned to the carbon to indicate the position of each substituent.
Branched Alkanes
1. Count the number of carbons in the longest chain of the substituent.
2. Numbering of the substituent chain starts from the carbon attached to the parent chain.
3. Substituents are listed in alphabetical order. To alphabetize, ignore numerical (di-, tri-, tetra-) prefixes (e.g., ethyl would come before dimethyl).
Cyclic Alkanes
1. The parent name is determined by the number of carbons in the largest ring (e.g., cycloalkane such as cyclopentane).
2. In the case where the ring is attached to a chain containing additional carbons, the ring is considered to be a substituent on the chain. A substituted ring that is a substituent on something else is named using the rules for branched alkanes.
3. The carbons of the ring are numbered such that the substituents are given the lowest possible numbers.
Straight Chain Alkanes
# Carbon Name Molecula Structural
Formula Formula
1 Methane CH4 CH4
2 Ethane C2H6 CH3CH3
3 Propane C3H8 CH3CH2CH3
4 Butane C4H10 CH3CH2CH2CH3
5 Pentane C5H12 CH3CH2CH2CH2CH3
6 Hexane C6H14 CH3(CH2)4CH3
7 Heptane C7H16 CH3(CH2)5CH3
8 Octane C8H18 CH3(CH2)6CH3
9 Nonane C9H20 CH3(CH2)7CH3
10 Decane C10H22 CH3(CH2)8CH3
ALKENE

In organic chemistry, an alkene, olefin, or olefine is an unsaturated chemical compound containing at least one carbon-to-carbon double bond. The simplest acyclic alkenes, with only one double bond and no other functional groups, form a homologous series of hydrocarbons with the general formula CnH2n.[2]
The simplest alkene is ethylene (C2H4), which has the International Union of Pure and Applied Chemistry (IUPAC) name ethene. Alkenes are also called olefins (an archaic synonym, widely used in the petrochemical industry). For bridged alkenes, the Bredt's rule states that a double bond cannot be placed at the bridgehead of a bridged ring system, unless the rings are large enough (8 or more atoms). Aromatic compounds are often drawn as cyclic alkenes, but their structure and properties are different and they are not considered to be alkenes.
Like single covalent bonds, double bonds can be described in terms of overlapping atomic orbitals, except that, unlike a single bond (which consists of a single sigma bond), a carbon-carbon double bond consists of one sigma bond and one pi bond. This double bond is stronger than a single covalent bond (611 kJ/mol for C=C vs. 347 kJ/mol for C—C) and also shorter with an average bond length of 1.33 Angstroms(133 pm).
-Matthew Reinier W. Garcia
Group 5 IV-Zara
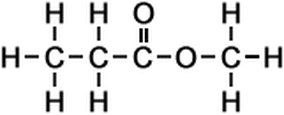
ESTER
What do you think about Ester? Is she a person? Some kind of new found element? But if you’re thinking it’s a person or element, then you’re wrong and this is kind of chemical compounds in organic compounds. In this article, we are going to explore the topics in Ester like how we’re going to name and draw their structures.
We are going to discuss first about the Ester group. Esters are chemical compounds consisting of a carbonyl adjacent to an ether linkage. They are derived by reacting an oxoacid with a hydroxyl compound such as an alcohol or phenol. They are usually derived from an inorganic acid or organic acid in which at least one -OH (hydroxyl) group is replaced by an -O-alkyl (alkoxy) group, and most commonly from carboxylic acids and alcohols. Esters are found in most naturally occurring fats and oils like the fatty acid esters of glycerol. Esters with low molecular weight are commonly used as fragrances and found in essential oils and pheromones. Phosphoesters form the backbone of DNA molecules. Nitrate esters, such as nitroglycerin, are known for their explosive properties, while polyesters are important plastics, with monomers linked by ester moieties. Natural occurring esters of organic acids in fruits and flowers give them their distinctive odors. It is also used for food aroma and taste, perfumes, synthetic fibres, and solvents.
In naming an Ester group, we must follow strictly the rules and steps in naming them. Its general formula where we can easily identify if it’s an Ester is R1CO2R2 (where R1 and R2 are alkyl groups). These are the steps to be followed in naming an Ester group.
Examples:
Name the structures below:
We are going to discuss first about the Ester group. Esters are chemical compounds consisting of a carbonyl adjacent to an ether linkage. They are derived by reacting an oxoacid with a hydroxyl compound such as an alcohol or phenol. They are usually derived from an inorganic acid or organic acid in which at least one -OH (hydroxyl) group is replaced by an -O-alkyl (alkoxy) group, and most commonly from carboxylic acids and alcohols. Esters are found in most naturally occurring fats and oils like the fatty acid esters of glycerol. Esters with low molecular weight are commonly used as fragrances and found in essential oils and pheromones. Phosphoesters form the backbone of DNA molecules. Nitrate esters, such as nitroglycerin, are known for their explosive properties, while polyesters are important plastics, with monomers linked by ester moieties. Natural occurring esters of organic acids in fruits and flowers give them their distinctive odors. It is also used for food aroma and taste, perfumes, synthetic fibres, and solvents.
In naming an Ester group, we must follow strictly the rules and steps in naming them. Its general formula where we can easily identify if it’s an Ester is R1CO2R2 (where R1 and R2 are alkyl groups). These are the steps to be followed in naming an Ester group.
- First, look for the alkane group attached with the double bond atom of oxygen and use it as a parent name for your structure.
- Then, find if there are other alkyl groups attached to the second oxygen atom.
- Lastly, change the name of the alkane by subtracting the letter “e” in the suffix –ane. (Example: –ane + -oate = anoate)
Examples:
Name the structures below:
|
Draw the following Esters:
|
- Donna Clarisse L. Calma
IV-Zara, Group 5
References:
http://chemwiki.ucdavis.edu/Organic_Chemistry/Esters/Nomenclature_of_Esters
http://www.angelfire.com/bc2/OrgChem/esters.html
http://en.wikipedia.org/wiki/Ester#IUPAC_nomenclature_of_Esters
http://www.ivy-rose.co.uk/Chemistry/Organic/Naming-Esters.php
IV-Zara, Group 5
References:
http://chemwiki.ucdavis.edu/Organic_Chemistry/Esters/Nomenclature_of_Esters
http://www.angelfire.com/bc2/OrgChem/esters.html
http://en.wikipedia.org/wiki/Ester#IUPAC_nomenclature_of_Esters
http://www.ivy-rose.co.uk/Chemistry/Organic/Naming-Esters.php
Nitrogen containing functional Group
Amines
|
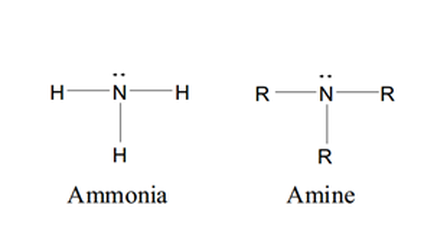
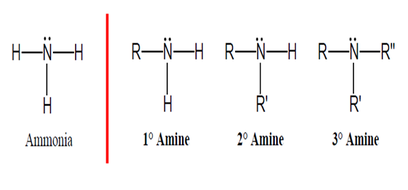
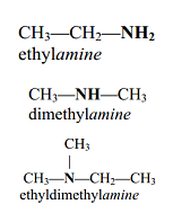
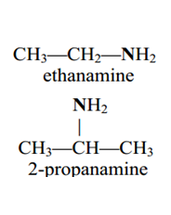
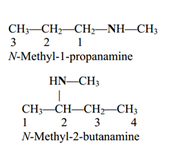
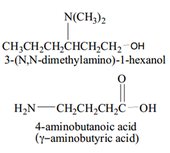
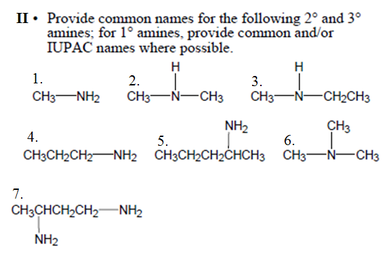
• Amines and amides are abundant in nature. They
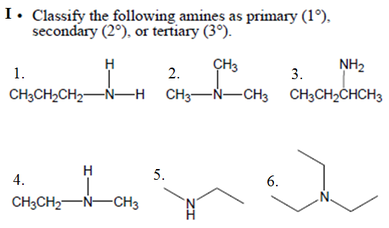
are a major component of proteins and enzymes, nucleic acids, alkaloid drugs, etc. • Amines are organic derivatives of ammonia, NH3, in which one or more of the three H’s is replaced by a carbon group. • Amines are classified as primary (1°), secondary (2°), or tertiary (3°), depending on how many carbon groups are connected to the nitrogen atom. |
Answer Key
I. 1. Primary 2. Tertiary 3. Primary 4. Secondarym 5. Secondary 6. Tertiary
II. 1. Methylamine 2. N-methyl -methanamine 3. N-methyl-ethanamine 4. Propylamine 5. Pentylamine
6. N,N-dimethyl-methanamine 7. 1,3-propyldiamine
I. 1. Primary 2. Tertiary 3. Primary 4. Secondarym 5. Secondary 6. Tertiary
II. 1. Methylamine 2. N-methyl -methanamine 3. N-methyl-ethanamine 4. Propylamine 5. Pentylamine
6. N,N-dimethyl-methanamine 7. 1,3-propyldiamine
- John Bennedick B. Quijano
IV-Zara, Group 5
IV-Zara, Group 5
Carboxylic Acid and Derivatives
|
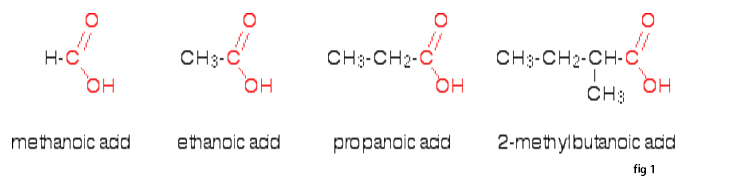
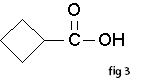
Carboxylic acids are the most acidic of the common organic functional groups. The general formula of a carboxylic acid is R-COOH, where R is some monovalent functional group. A carboxyl group (or carboxy) is a functional group consisting of a carbonyl (RR'C=O) and a hydroxyl (R-O-H), which has the formula -C(=O)OH, usually written as –COOH.
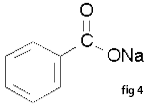
Carboxylic acids are polar. Because they are both hydrogen-bond acceptors (the carbonyl) and hydrogen-bond donors (the hydroxyl), they also participate in hydrogen bonding. Together the hydroxyl and carbonyl group forms the functional group carboxyl. Examples of Carboxylic Acid: (fig 1) The name counts the total number of carbon atoms in the longest chain - including the one in the -COOH group. If you have side groups attached to the chain, notice that you always count from the carbon atom in the -COOH group as being number 1. Nomenclature of Carboxylic Acids a. Carboxylic acids are named by dropping the -e ending of the parent name and adding -oic acid. The substituent name for a -COOH group is carboxy. pentanoic acid or 1-carboxybutane (fig 2) b. Ring compounds. When one or more –COOH groups are attached to a ring, the ring is named followed by the word -carboxylic acid. The carbon atom to which the -COOH group is attached is numbered 1 and substituents are numbered relative to that number. cyclobutanecarboxylic acid(fig 3) c. Salts of carboxylic acids are named by first naming the metal, followed by a space and then by changing the name of the acid by dropping the -oic acid ending and adding -oate. sodium benzoate(fig 4) Derivatives of Carboxylic Acids The important classes of organic compounds known as alcohols, phenols, ethers, amines and halides consist of alkyl and/or aryl groups bonded to hydroxyl, alkoxyl, amino and halo substituents respectively. If these same functional groups are attached to an acyl group (RCO–) their properties are substantially changed, and they are designated as carboxylic acid derivatives. Carboxylic acids have a hydroxyl group bonded to an acyl group, and their functional derivatives are prepared by replacement of the hydroxyl group with substituents, such as halo, alkoxyl, amino and acyloxy. Nomenclature Three examples of acyl groups having specific names were noted earlier. These are often used in common names of compounds. In the following examples the IUPAC names are color coded, and common names are given in parentheses. • Esters: The alkyl group is named first, followed by a derived name for the acyl group, the -oic or -ic suffix in the acid name is replaced by ate. e.g. CH3(CH2)2CO2C2H5 is ethyl butanoate (or ethyl butyrate). Cyclic esters are called lactones. A Greek letter identifies the location of the alkyl oxygen relative to the carboxyl carbonyl group. • Acid Halides: The acyl group is named first, followed by the halogen name as a separate word. e.g. CH3CH2COCl is propanoyl chloride (or propionyl chloride). • Anhydrides: The name of the related acid(s) is used first, followed by the separate word "anhydride". e.g. (CH3(CH2)2CO)2O is butanoic anhydride & CH3COOCOCH2CH3 is ethanoic propanoic anhydride (or acetic propionic anhydride). • Amides: The name of the related acid is used first and the oic acid or ic acid suffix is replaced by amide (only for 1º-amides). e.g. CH3CONH2 is ethanamide (or acetamide). 2º & 3º-amides have alkyl substituents on the nitrogen atom. These are designated by "N-alkyl" term(s) at the beginning of the name. e.g. CH3(CH2)2CONHC2H5 is N-ethylbutanamide; & HCON(CH3)2 is N,N-dimethylmethanamide (or N,N-dimethylformamide). Cyclic amides are called lactams. A Greek letter identifies the location of the nitrogen on the alkyl chain relative to the carboxyl carbonyl group. • Nitriles: Simple acyclic nitriles are named by adding nitrile as a suffix to the name of the corresponding alkane (same number of carbon atoms). Chain numbering begins with the nitrile carbon . Commonly, the oic acid or ic acid ending of the corresponding carboxylic acid is replaced by onitrile. A nitrile substituent, e.g. on a ring, is named carbonitrile. e.g. (CH3)2CHCH2C≡N is 3-methylbutanenitrile (or isovaleronitrile). -Christine Joy G. Jimenez Group 5 IV-Zara |
ALCOHOLS
A functional group is a particular arrangement of a few atoms that imparts certain characteristics properties to organic molecules. For example, alcohols are derivatives of hydrocarbons in which one or more of the hydrogen atoms has been replaced by a hydroxyl (OH) functional group.
In the IUPAC system, alcohols are named for the longest continuous chain containing the –OH group, with that functional group having the lowest numbers. The ending –e is changed to –ol to indicate the alcohol functional group. Thus, the alcohol derived from methane is methanol, and the alcohol derived from ethane is ethanol.
In the common system, alcohols are named by taking the name of the alkyl group and adding the word alcohol. Example of the name of simple alcohols is Ethyl Alcohol.
What is ETHYL ALCOHOL?
Ethyl alcohol (ethanol), CH3CH2OH, is known commonly as grain alcohol. In the hospital the word alcohol means ethyl alcohol.
One important property of ethyl alcohol is its ability to denature protein. Because of this property, ethyl alcohol is widely used as an antiseptic.
As an antiseptic, 70 percent alcohol is preferred to a stronger solution. It would seem that if 70 percent alcohol is a good antiseptic, then 100 percent alcohol would be even better; however the reverse is true. The 70 percent alcohol is actually a better antiseptic than the 100 percent alcohol.
Pure alcohol coagulates protein on contact. Suppose the pure alcohol is poured over a single-celled organism. The alcohol will penetrate the cell wall of that organism in all directions, coagulating the protein just inside the cell wall. The ring of coagulated protein would then prevent the alcohol from penetrating farther into a cell, and no more coagulation would take place. At that time the cell would become dormant, but not dead. Under the proper conditions the organism could again begin to function. If 70 percent alcohol is poured over a single-celled organism, the diluted alcohol also coagulates the protein, but a slower rate, so that it penetrates all the way through the cell before coagulation can block it. Then all the cell proteins are coagulated, and the organism dies.
Ethyl alcohol can also be used for sponge baths to reduce the fever of a patient. When alcohol is placed on the skin it evaporates rapidly. In order to evaporate, alcohol requires heat. This heat comes from the patient’s skin. Thus, an alcohol sponge bath will remove heat from patient’s skin and so slower the body temperature. A water sponge bath will do the same thing, but water evaporates more slowly than alcohol so the heat is removed more slowly. However, water sponge baths are in common use in many hospitals because they are cheaper. For children it is kinder to allow them to play in a tepid bath. The alcohol sponge bath cools them too rapidly is uncomfortable. Since alcohol is flammable it cannot be used in room where oxygen is in use.
Alcohol is used as a solvent for many substances. Alcohol solutions are called tinctures. Tincture iodine consists of iodine dissolved in alcohol.
Ethyl alcohol is also used as beverage. The concentration of alcohol in alcoholic beverages is expressed as “proof”. The proof is twice the percentage of alcohol in the solution. Thus a beverage marked “100 proof” contains 50 percent alcohol. Alcohol slows reaction time, so driving under the influence of alcohol can be very dangerous. Alcohol is not a stimulant; it actually depresses the nervous system and can remove an individual’s normal inhibitions. Excessive use of alcohol may cause the destruction of the liver, a condition known as cirrhosis.
Alcohol can be prepared from the fermentation of blackstrap molasses, the residues that result from the purification of cane sugar. The principal constituent of molasses is sucrose (C12H22 O11). The fermentation reaction is brought about by enzymes present in yeast.
The starches present in grains can be converted into a sugar by malt. The sugar thus produced can be fermented under the influence of the enzymes in yeast to yield ethyl alcohol. Hence ethyl alcohol is also known as grain alcohol.
Reference:
http://en.wikibooks.org/wiki/Organic_Chemistry/Aromatics
http://www.hz.genium.com
-Camille R. Villania
Group 5 IV Zara